Experimental compound kills cancer, spares immune cells
UT Southwestern scientists identify N6F11 as promising treatment to target various cancers
UT Southwestern Medical Center researchers have identified a compound that selectively eliminates cancer cells while sparing immune cells in a form of cell death known as ferroptosis. The findings, published in Science Translational Medicine, could lead to new treatments for a wide variety of cancer types, the study authors say.

Daolin Tang, M.D., Ph.D., is Professor of Surgery and a member of the Harold C. Simmons Comprehensive Cancer Center at UT Southwestern.
“Our study is the first to report a cancer cell-specific induction of ferroptosis,” said Daolin Tang, M.D., Ph.D., Professor of Surgery at UT Southwestern, who co-led the study with Rui Kang, M.D., Ph.D., Associate Professor of Surgery at UT Southwestern. Drs. Tang and Kang are members of the Harold C. Simmons Comprehensive Cancer Center.
Just over a decade ago, researchers elsewhere discovered ferroptosis, a type of cell death characterized by an accumulation of large amounts of iron and uncontrolled lipid peroxidation. Because some types of cancer develop resistance to chemotherapy drugs, finding ways to prompt ferroptosis quickly became the focus of significant cancer research, Dr. Tang explained.
Most of these efforts focused on inhibiting an antioxidant enzyme called GPX4, a master repressor of ferroptosis. However, Dr. Tang said, GPX4-inhibiting compounds identified thus far not only induced ferroptosis in cancer cells but also in a variety of immune cell types – causing side effects in lab animals while also hindering the immune system’s natural anti-cancer response.

Rui Kang, M.D., Ph.D., is Associate Professor of Surgery and a member of the Harold C. Simmons Comprehensive Cancer Center at UT Southwestern.
Searching for a compound that targets GPX4 only in cancer cells, Drs. Tang and Kang and their colleagues screened more than 4,200 compounds from a pharmaceutical company’s drug library, dosing human pancreatic cancer cells growing in petri dishes. These experiments found that a compound called N6F11 not only reduced the amount of GPX4 present in cells, but also readily caused these cells to die from ferroptosis. Nearly half of the cells were dead within 12 hours. The researchers had similar results when they dosed other cancer cell types, including bladder, breast, and cervical, with N6F11.
Additional experiments showed that N6F11 binds to another protein called TRIM25, which regulates the amount of GPX4 and other proteins and is especially abundant in cancer cells. That binding prompts TRIM25 to tag GPX4 with ubiquitin, a protein that directs GPX4 to cellular garbage dumps where it’s degraded. With GPX4 gone, cells die from ferroptosis.
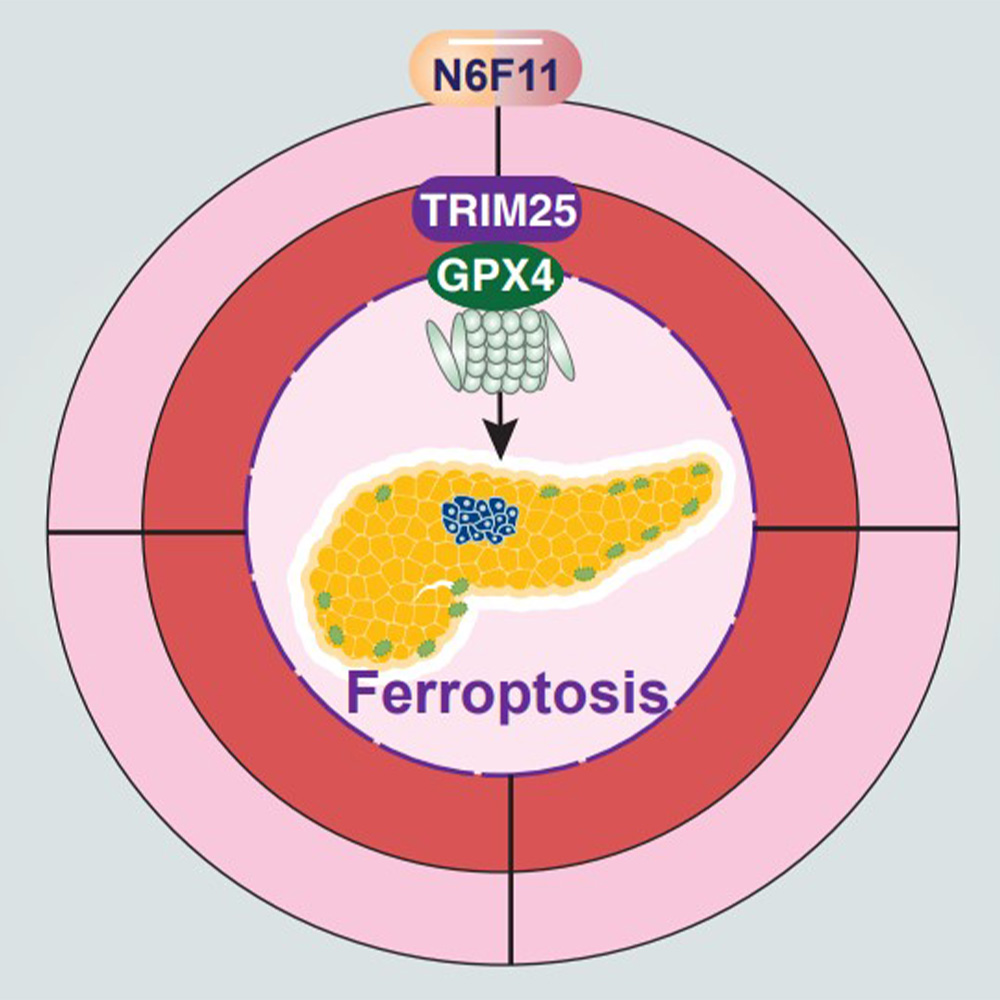
UT Southwestern researchers have found a compound called N6F11 that triggers selective induction of ferroptosis in cancer cells to suppress pancreatic tumor growth.
Further testing on mouse models with pancreatic cancer showed N6F11 not only virtually stopped cancer progression, Dr. Tang said, but it also appeared to be nontoxic for healthy tissues or cells. The immune-inhibiting side effects seen in previous research focused on inhibiting GPX4 were absent with N6F11. In the next phase, researchers gave animals a combination of N6F11 and a cancer treatment known as an immune checkpoint inhibitor that is commonly used in patients. This combination significantly improved their survival rates compared to mice that received either treatment separately.
Future research will focus on developing N6F11 into a viable drug – a process that could take several years – as well as searching drugs already approved by the U.S. Food and Drug Administration for those that act similarly to N6F11, which would require less time and money, Dr. Tang said.
Other UTSW researchers who contributed to this study are Zhuan Zhou, Ph.D., Assistant Professor of Dermatology, and Chunhua Yu, M.D., Ph.D., Manager of the Tang Lab.
This study was funded by National Institutes of Health grants (R01CA160417, R01CA229275, and R01CA211070).
